Introduction
Pressure ulcers (PUs) are a significant and growing public health concern, particularly among aging and medically complex populations receiving care in post-acute settings. Patients in these environments frequently experience reduced mobility, impaired tissue perfusion, and multiple comorbidities that contribute to prolonged healing trajectories and increased risk of wound chronicity.1,2 At a biological level, chronic PUs are often characterized by a stalled inflammatory state marked by excessive protease activity, impaired extracellular matrix deposition, and delayed re-epithelialization, all of which impede progression toward durable closure.3,4
In response to the therapeutic challenges posed by chronic and advanced-stage PUs, clinicians have increasingly adopted advanced wound therapies designed to actively support tissue repair processes. Among these, biologically active wound matrices represent an evolving class of treatments intended to modulate the wound microenvironment. These matrices aim to promote granulation tissue formation, regulate inflammation, and facilitate epithelial migration in ulcers that have failed to progress with standard care alone.5,6
Human keratin matrices (HKMs) represent a distinct category within this broader class of biologically active wound biomaterials. Keratin is a structural protein that constitutes a major component of the epidermis and its appendages and is characterized by extensive crosslinking that confers resistance to proteolytic degradation.7 Preclinical and translational studies have demonstrated that keratin-based biomaterials influence multiple cellular processes relevant to wound repair, including keratinocyte activation and migration, fibroblast proliferation, and modulation of inflammatory signaling pathways.7,8 In contrast to collagen-based matrices, which may be susceptible to rapid degradation in protease-rich chronic wound environments such as advanced PUs, keratin matrices appear to resist degradation while remaining biologically interactive.9
Emerging mechanistic evidence suggests that keratin matrices may address key drivers of PU chronicity. Experimental studies have shown that keratin biomaterials can attenuate elevated matrix metalloproteinase activity, including suppression of matrix metalloproteinase-9 through zinc ion chelation, a mechanism particularly relevant in chronic PUs characterized by excessive protease burden.10,11 Additional work indicates that keratin-based materials may promote a shift in macrophage phenotype toward anti-inflammatory and pro-regenerative states, potentially facilitating resolution of prolonged inflammation and progression toward tissue repair.8,12
Clinical studies have reported favorable healing outcomes associated with HKM use across multiple chronic wound types. Multicenter trials and case series have demonstrated improved healing rates in diabetic foot ulcers, venous leg ulcers, and other difficult-to-heal lower extremity wounds treated with HKMs in conjunction with standard care.13-16
However, PUs remain underrepresented in controlled clinical trials, and existing studies are often conducted in narrowly defined populations. As a result, their findings may not fully reflect the clinical heterogeneity, care complexity, and treatment variability encountered in routine post-acute PU management.
Real-world data sources, including large wound registries, provide an opportunity to evaluate treatment effectiveness for PUs across diverse care environments and patient populations. When paired with analytical approaches designed to address confounding and treatment selection bias, real-world evidence can complement other forms of clinical evidence by offering insight into how advanced wound therapies perform under typical conditions of use.17,18 Bayesian analytical frameworks are particularly well suited to this context, as they enable direct probabilistic interpretation of treatment effects while formally quantifying uncertainty in observational settings.19
Given these considerations, evaluating the real-world effectiveness of HKMs specifically for PU healing in post-acute care environments is warranted. This study aims to assess the impact of HKM use on PU healing outcomes compared with standard of care (SOC) alone among patients treated in post-acute settings. Using observational registry data and a Bayesian analytical approach, the analysis seeks to estimate both the probability and magnitude of treatment benefit associated with HKM use in real-world clinical practice.
Methods and materials
Data source and study population
This study analyzed deidentified real-world pressure ulcer (PU) data derived from a national post-acute wound registry capturing longitudinal PU data from long-term acute care hospitals and skilled nursing facilities to support longitudinal surveillance of prevalence, treatment patterns, and healing outcomes in post-acute care environments across an extended observational period. The registry aggregates PU-level and patient-level data contributed by a national network of participating post-acute care providers, including long-term acute care hospitals, skilled nursing facilities, and affiliated sub-acute institutions.
Clinical data are extracted directly from partner electronic medical record (EMR) systems and processed through automated ingestion and harmonization pipelines prior to analysis. Data are standardized using a common data model that captures detailed PU characteristics (including anatomical location, stage, size, and depth), patient demographics, documented comorbidities, and elements of ulcer management. Unique PU identifiers enable longitudinal linkage of repeated assessments, allowing ulcers to be followed across multiple encounters to evaluate healing trajectories, stage transitions, and closure events over time.
All data within the LiftOff Registry are fully de-identified before analysis and stored in a secure, HIPAA-compliant environment. The registry operates under a centralized governance framework that provides oversight for data integrity, ethical use, and authorization of secondary analyses for research and quality improvement. Use of registry data for this study was approved under the registry’s governance and data-use policies. Formal Institutional Review Board review was not required due to the retrospective analysis of deidentified data.
Study population
The analytic cohort included adult patients (≥18 years) treated between March 2002 and January 2025 who had at least one documented PU and sufficient longitudinal follow-up to assess healing status. PUs were excluded if key baseline variables required for matching or outcome modeling were missing. Baseline covariates were measured at PU identification, prior to subsequent HKM initiation. Baseline variables included patient age, sex, diabetes status, baseline PU area, and PU stage at identification.
Healing was defined as complete PU closure or re-epithelialization as documented in the clinical record by the treating provider. Longitudinal PU identifiers were used to verify closure events across successive clinical encounters, minimizing misclassification due to transient documentation changes.
The treatment group consisted of pressure ulcers that received a HKM as part of routine clinical care, in addition to SOC. HKM exposure was defined as any documented application of the product; no minimum number of applications was required for inclusion. Comparator PUs were managed with SOC alone and did not receive HKM during the observed treatment period. SOC included routine PU management practices as determined by the treating facility and clinician and may have varied across sites.
Analysis
This observational comparative effectiveness study evaluated PU healing outcomes among patients receiving either SOC alone or SOC combined with HKM. Bayesian propensity score methods were applied to reduce confounding and improve balance between treatment groups prior to estimation of treatment effects on PU healing outcomes.
The index date for all analyses was defined as the date of PU identification (first recorded clinical encounter). Propensity scores were estimated using baseline covariates measured prior to any HKM exposure, reflecting the counterfactual probability of receiving HKM versus SOC under real-world treatment selection.
All Bayesian models were implemented in Python 3.12 using the probabilistic programming library PyMC (version 5.23.0). The Bayesian Model-Building Interface (version 0.15.0) was used for model specification, and ArviZ (version 0.21.0) was employed for posterior diagnostics and visualization.
Bayesian methods were selected to provide a coherent probabilistic framework for estimating treatment effects while propagating uncertainty across both the matching and outcome-modeling stages. Unlike frequentist approaches that yield point estimates and confidence intervals, Bayesian inference generates full posterior distributions that directly quantify uncertainty in treatment assignment and outcome estimation, allowing this uncertainty to be carried forward into causal effect inference.17-19 Prior work has demonstrated the advantages of Bayesian propensity score modeling for observational causal inference, particularly in settings characterized by non-random treatment assignment and heterogeneous populations.19
All regression coefficients were assigned weakly informative priors (Normal [0,1]), with a Normal (0,1.5) prior specified for the intercept. Four Markov chain Monte Carlo chains were run with 1,000 warm-up iterations and 1,000 posterior draws per chain using the No-U-Turn Sampler.
Posterior mean propensity scores were used to perform 1:1 nearest-neighbor matching without replacement, with exact matching enforced for categorical covariates and nearest matching applied to continuous variables. A single replacement was permitted only when no alternative match was available. Following matching, covariate balance was assessed using visual inspection of empirical cumulative distribution functions and summary baseline characteristics (including standardized differences). Within the matched cohort, the probability of PU healing was modeled using Bayesian logistic regression with study arm and baseline PU stage specified as predictors. Posterior predictive probabilities of healing were estimated for each study arm, and the treatment effect was summarized as a posterior risk ratio (RR), calculated as the ratio of modeled healing probabilities between HKM and SOC for each posterior draw. Results are reported as posterior mean RR with 95% highest density intervals (HDI) and posterior probability of benefit (RR > 1). Because logistic regression estimates effects on the log-odds scale, risk ratios were derived from posterior predictive probabilities rather than from exponentiated regression coefficients.
Time to first HKM application was defined as the interval between PU identification and first recorded HKM application. Time under HKM treatment was defined as the total treatment span from first to last recorded application. These timing variables were summarized descriptively using cumulative distribution functions.
Results
Cohort description and matching
Before matching, the SOC cohort included 9356 patients with 14,740 PUs, of which 33.2% achieved healing. The HKM cohort included 58 patients with 81 PUs, with a crude healing rate of 54.3%.
After Bayesian propensity score matching, 161 PUs were retained for analysis, including 81 HKM-treated PUs and 80 SOC-treated PUs. Overall descriptive characteristics of the matched cohorts, including wound size at identification and wound size at the final recorded assessment, are summarized in Table 1. Patients in both groups were similar with respect to age and diabetes prevalence. Median PU area at identification was larger in the SOC group than in the HKM group, while end-of-study median PU area was smaller than baseline in both groups, consistent with wound progression over time.
TABLE 1 Descriptive summary of matched pressure ulcer cohorts at identification and end of follow-up
| Study arm | Ulcers (n) | Mean age | Diabetes (%) | Starting median area (cm2) | Ending median area (cm2) | Healed (%) | Mean follow-up time (weeks) |
|---|---|---|---|---|---|---|---|
| SOC | 80 | 64.2 | 15.0 | 43.0 | 12.0 | 33.8 | 16.9 |
| HKM | 81 | 66.1 | 14.8 | 27.8 | 10.6 | 54.3 | 38.1 |
SOC, standard of care; HKM, human keratin matrix
Covariate balance after matching
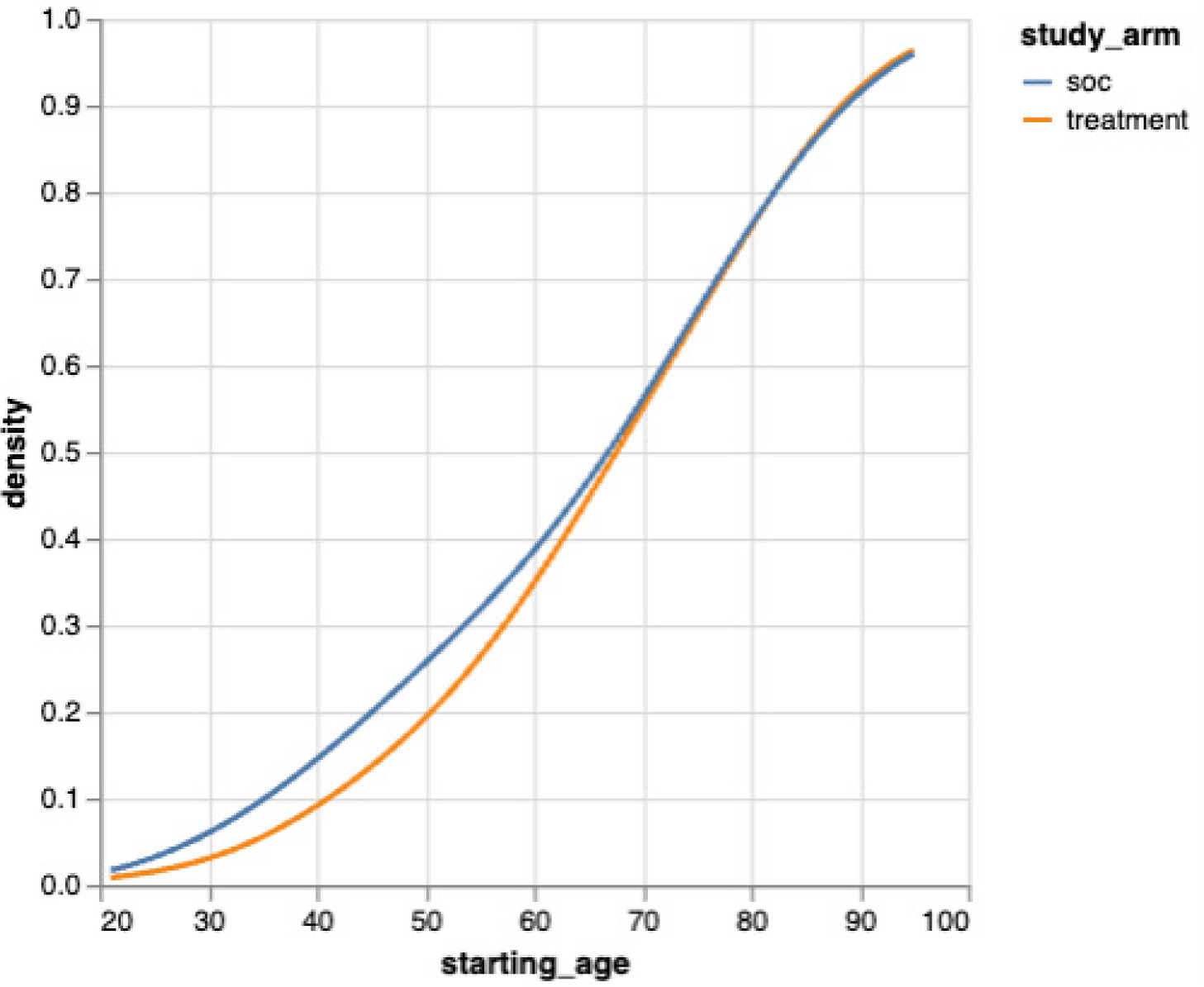
Covariate balance following propensity score matching is illustrated using empirical cumulative distribution functions for baseline PU area (Figure 1) and patient age (Figure 2), demonstrating substantial overlap between study arms.
FIGURE 1 Empirical cumulative distribution of baseline pressure ulcer area after matching.
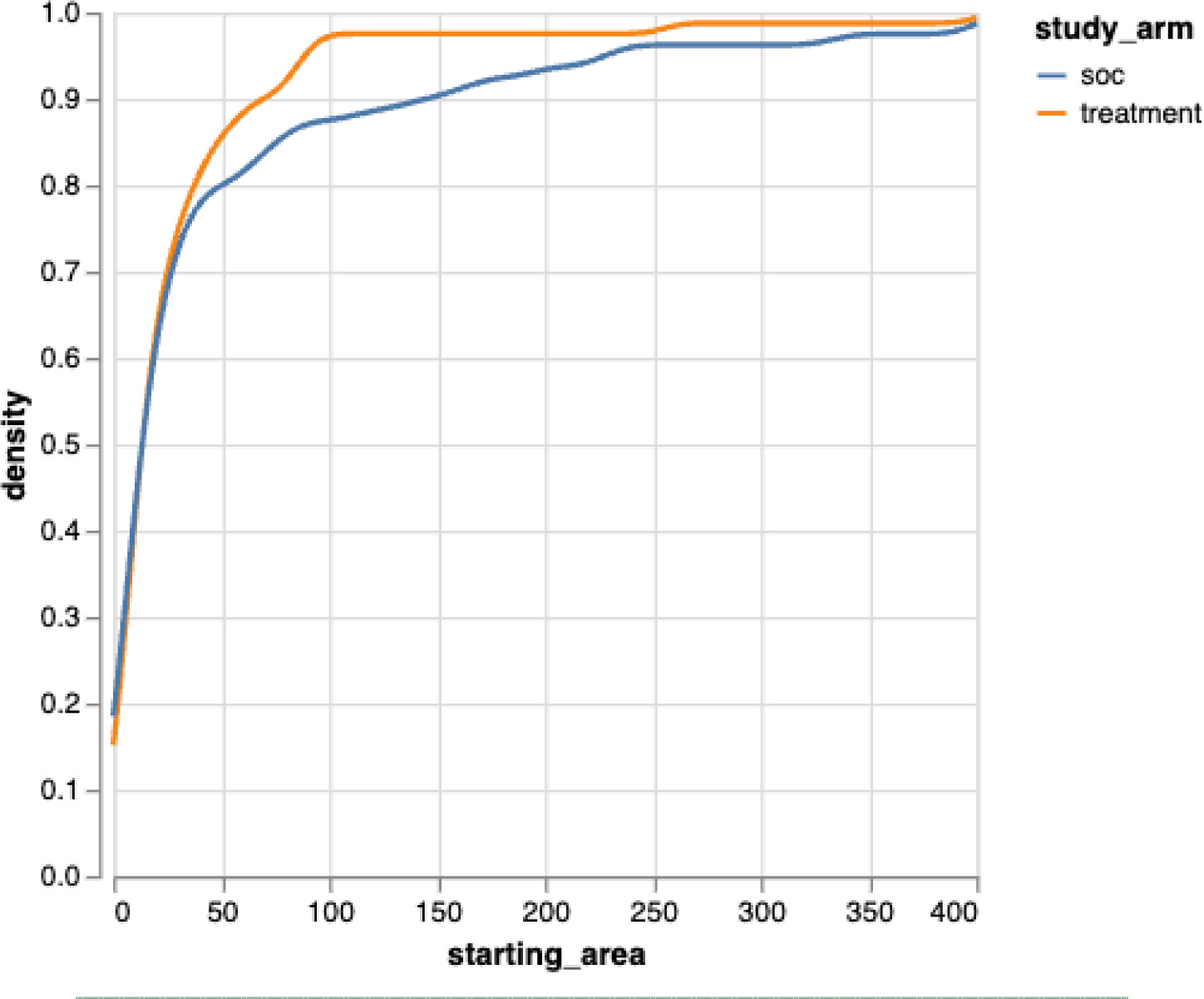
FIGURE 2 Empirical cumulative distribution of patient age after matching.
Treatment timing and exposure
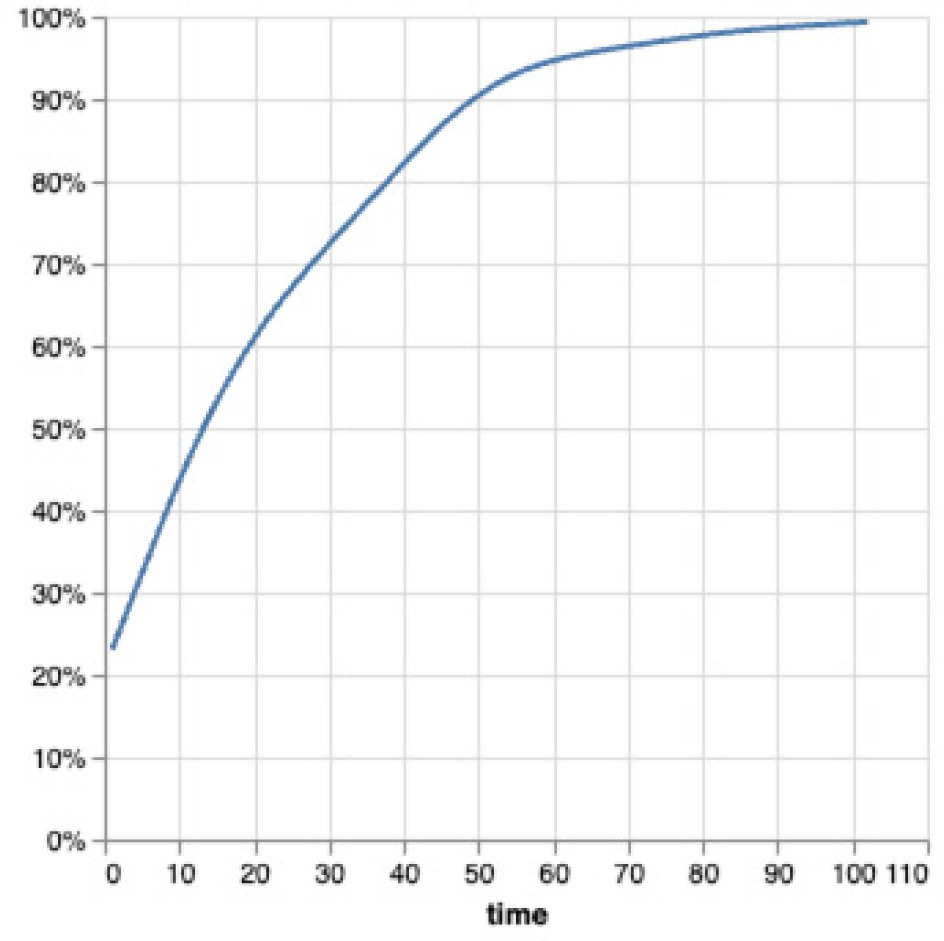
Among HKM-treated PUs, time to first HKM application varied substantially. Cumulative distribution analysis demonstrated that HKM was typically introduced after an initial period of SOC management rather than immediately at PU identification (Figure 3). The distribution indicates that HKM was generally initiated several weeks after identification rather than at the time of initial diagnosis.
FIGURE 3 Empirical cumulative distribution of time to first HKM application (weeks) following pressure ulcer identification.
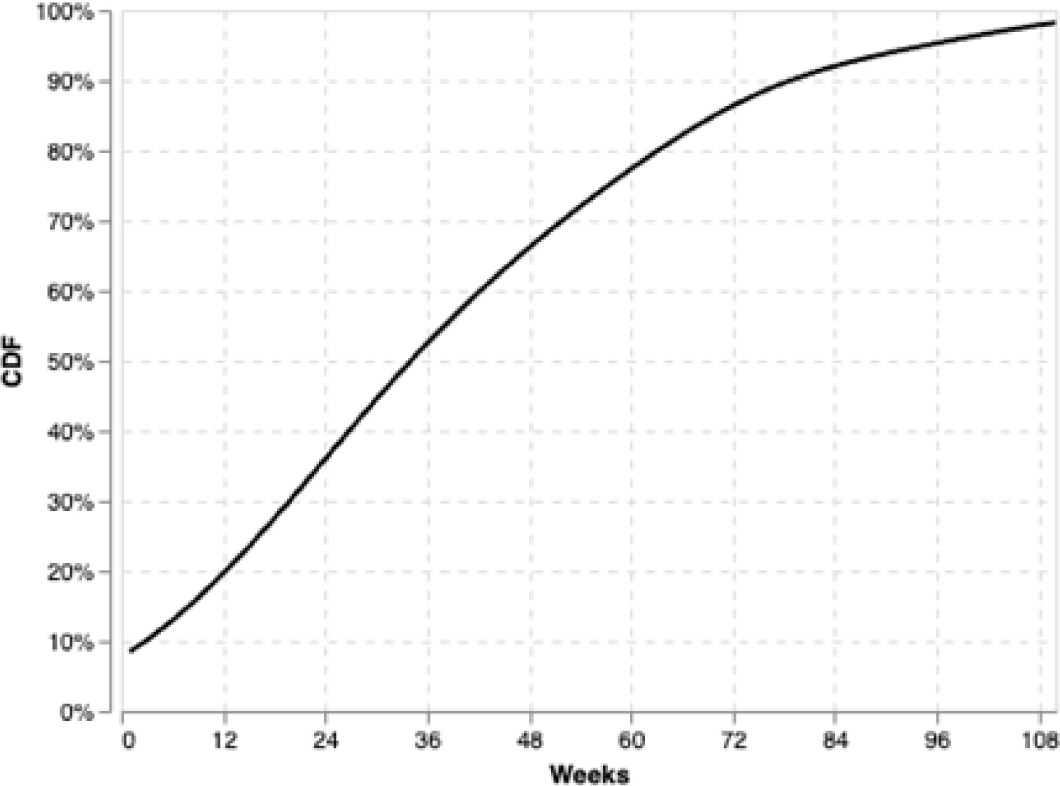
The duration of HKM exposure was similarly heterogeneous. Cumulative distribution analysis of time under HKM treatment—defined as the total treatment span from first to last application—demonstrated a wide range of exposure durations, with many PUs receiving HKM over extended intervals (Figure 4). Despite the extended duration of application for some patients, the average number of HKM applications among treated PUs was approximately 6.5. This reflects heterogeneity in frequency of application as well as the length of the treatment course.
FIGURE 4 Duration of HKM treatment exposure.
Modeled healing outcomes and treatment effect
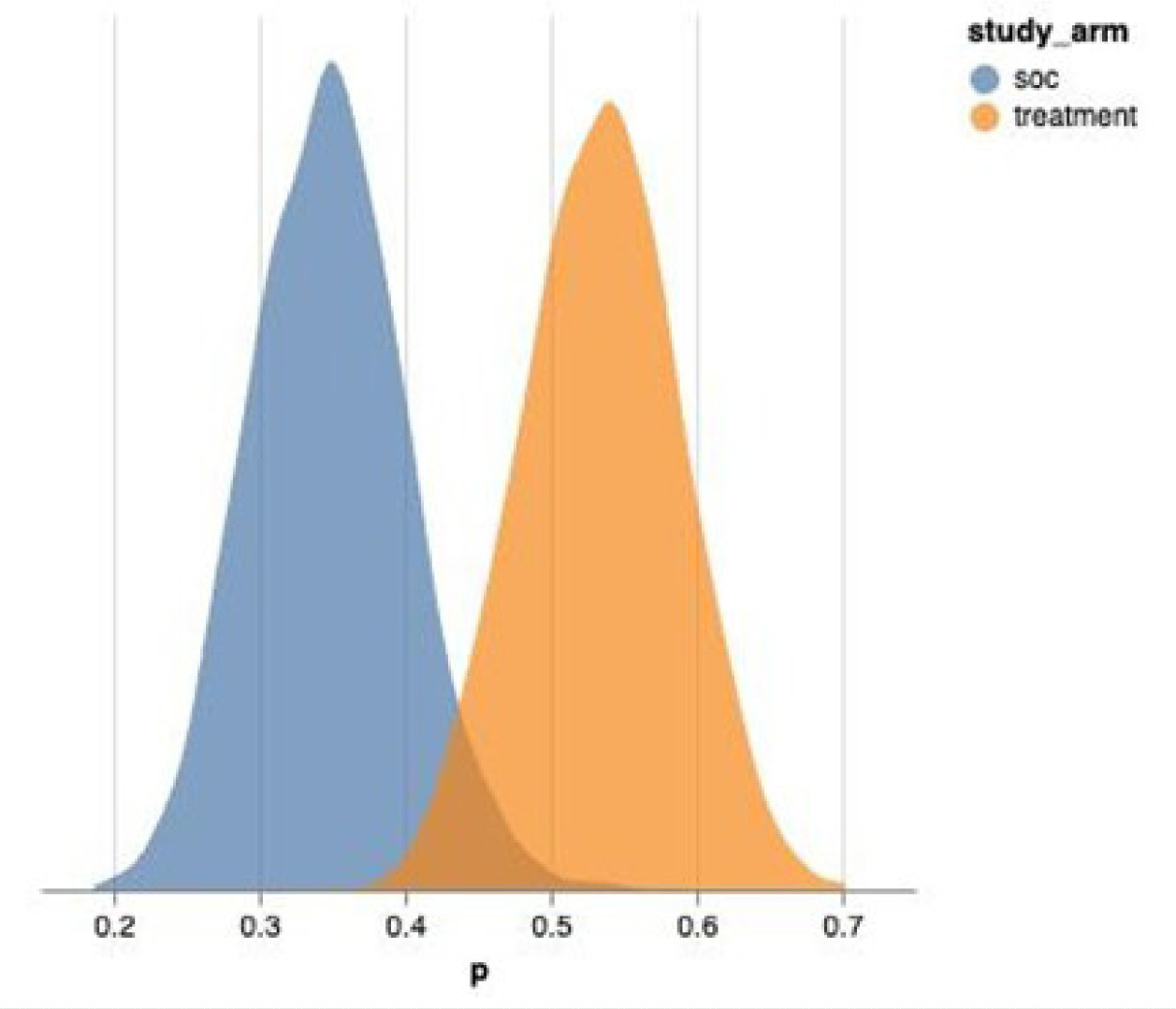
Posterior distributions of the modeled probability of healing demonstrated clear separation between study arms, with HKM-treated PUs exhibiting higher modeled healing probabilities compared with SOC alone (Figure 5). The posterior distribution for the HKM arm was consistently centered at a higher probability of healing relative to the SOC arm, indicating a treatment-associated shift in healing likelihood.
FIGURE 5 Posterior distributions of modeled healing probability by study arm.
The primary analysis estimated the effect of HKM treatment on PU healing using a Bayesian logistic regression model. Posterior draws yielded a posterior mean RR of approximately 1.6, indicating a higher modeled probability of healing among HKM-treated PUs compared with SOC alone (Figure 6).
FIGURE 6 Posterior distribution of the HKM treatment risk ratio (RR) for healing.
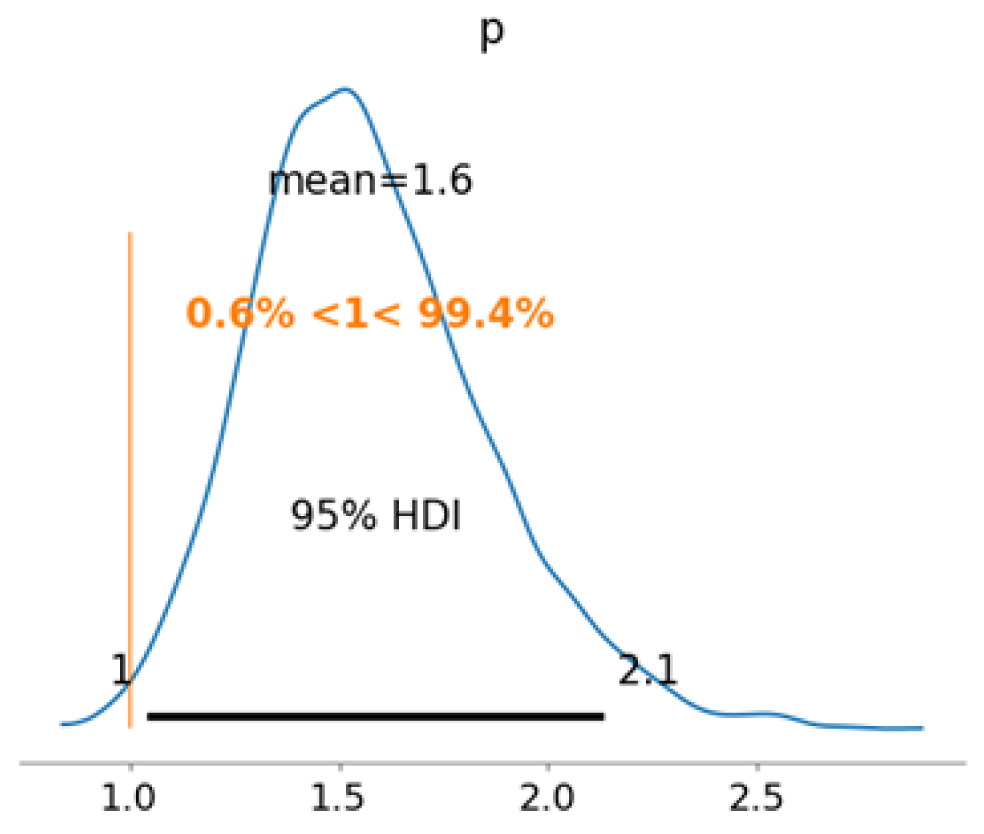
The posterior probability that the treatment effect exceeded unity (RR > 1) was approximately 99.4%, indicating strong posterior support for a beneficial association between HKM use and PU healing. The posterior interval from the updated analysis corresponds to a 95% HDI, spanning approximately 1.0 to 2.1. Model-implied healing probabilities by study arm and baseline PU stage are presented in Table 2.
TABLE 2 Model-implied probability of pressure ulcer healing by study arm and baseline stage
| Baseline PU Stage | SOC % (95% HDI) | HKM % (95% HDI) |
|---|---|---|
| Overall | 34.8% (23.3–47.6) | 53.6% (39.6–67.8) |
| Deep tissue injury | 40.3% (16.8–64.7) | 58.3% (34.4–80.7) |
| Stage 3 | 36.6% (21.9–51.9) | 55.2% (40.4–71.7) |
| Stage 4 | 34.1% (23.7–46.0) | 52.7% (41.1–64.8) |
| Unstageable | 28.4% (2.3–61.9) | 43.3% (8.6–80.3) |
Stage 2 pressure ulcers were excluded from comparative inference due to the absence of matched SOC comparators. PU, pressure ulcer; HDI, highest density interval; SOC, standard of care; HKM, human keratin matrix.
Discussion
This real-world observational analysis demonstrates a high posterior probability that treatment with HKM is associated with improved PU healing compared with SOC alone in post-acute care settings. The posterior mean risk ratio was approximately 1.6, corresponding to an estimated 60% greater modeled probability of healing compared with SOC. The posterior probability that the risk ratio exceeded 1 was greater than 99%, with the posterior distribution consistently centered above unity. These findings suggest a positive association between HKM use and PU healing in a clinically complex population representative of routine post-acute PU care.
The observed treatment effect was evident across multiple advanced ulcer stages, including deep tissue injuries, Stage 3 PUs, Stage 4 PUs, and unstageable PUs. This is particularly notable given the well-documented challenges associated with healing advanced-stage PUs in post-acute environments, where patients often present with impaired mobility, multiple comorbidities, and prolonged wound duration.1-4 Also notable, many of the HKM patients had already been treated with SOC for extended periods without healing. The consistency of the modeled benefit across severe PU categories suggests that HKM may be especially relevant for difficult-to-heal PUs commonly encountered in post-acute settings.
Unlike prior real-world evaluations that have examined heterogeneous classes of advanced wound products, this study focused specifically on a single biomaterial category. This narrower exposure definition reduces interpretive ambiguity and allows for more precise estimation of treatment effects attributable to HKM. The findings are consistent with prior clinical and translational studies demonstrating favorable healing outcomes associated with keratin-based biomaterials in diabetic foot ulcers and venous leg ulcers.13-16 Mechanistically, keratin matrices have been shown to resist proteolytic degradation and to modulate key biological processes involved in chronic PU pathophysiology, including excessive matrix metalloproteinase activity and dysregulated inflammation.7-12 The present analysis extends this evidence by demonstrating that these biologically plausible mechanisms translate into measurable healing benefits in real-world post-acute care practice.
The Bayesian analytical framework used in this study offers several advantages for interpreting treatment effects in observational datasets. Rather than relying on dichotomous hypothesis testing, Bayesian inference yields full posterior distributions that quantify uncertainty and allow direct probabilistic statements regarding treatment benefit.17-19 This approach is particularly valuable in real-world PU studies, where treatment assignment is non-random, sample sizes for specific therapies may be modest, and patient and ulcer heterogeneity is substantial.
Treatment timing analyses demonstrated substantial heterogeneity in real-world HKM use. Many PUs received HKM following an extended period of SOC management rather than early during non-healing. Similar patterns have been reported in prior real-world studies of advanced wound therapies, where delayed initiation may attenuate observed treatment effects relative to earlier use.13-15 Although this study was not designed to evaluate the causal impact of treatment timing, the observation that benefit was evident despite delayed initiation suggests that HKM may retain clinical utility even in PUs that have failed to respond to prolonged SOC.
Strengths and limitations
As with all observational studies, the potential for residual confounding remains despite the use of Bayesian propensity score matching to improve covariate balance. Unmeasured factors—such as PU chronicity prior to registry entry, clinician treatment preferences, or facility-level care practices—may have influenced treatment selection and outcomes. The relatively small number of HKM-treated PUs limited the precision of subgroup estimates for certain ulcer stages, particularly Stage 2 PUs, for which no matched SOC comparators were available.
Despite these limitations, this study has notable strengths. The use of a large, national real-world registry enhances generalizability to typical post-acute care populations, which are often underrepresented in randomized controlled trials. The Bayesian framework allowed uncertainty to be propagated through both the matching and outcome-modeling stages, providing a coherent and transparent estimate of treatment effect. The extended study period (2002–2025) further enabled capture of a broad spectrum of post-acute care practices and patient complexity while preserving internal validity through baseline covariate matching.
No safety or adverse event data were systematically captured in this registry. However, no HKM-related complications were identified in the available clinical documentation, consistent with prior reports describing favorable tolerability profiles for keratin-based wound matrices.13-16
Conclusion
In this real-world observational study of patients treated in post-acute care settings, use of a HKM was associated with a high posterior probability of improved PU healing compared with SOC alone. The modeled treatment effect favored HKM across multiple advanced PU stages, supporting its potential role as an adjunctive therapy for complex or non-healing PUs commonly encountered in post-acute environments.
These findings suggest that integration of HKM into post-acute PU management pathways may improve healing outcomes in medically complex populations. Further studies with larger cohorts and prospective designs may help refine estimates of treatment effect, clarify optimal timing of initiation, and identify patient subgroups most likely to benefit. Together, this analysis contributes meaningful real-world evidence supporting the clinical utility of keratin-based matrices in routine post-acute PU care.